Sustainable fuels
Green fuels for a clean future in energy and propulsion.
Green fuels as the key to clean energy and propulsion
Sustainable fuels are key to a carbon-neutral future for energy and propulsion. We rely on HVO, hydrogen, and on other e-fuels produced using green, renewable electricity. This way, our combustion engines can provide clean energy for today and tomorrow.
Hydrogen - we have the whole picture in mind
In future, electricity will be generated from renewables – for example with the aid of solar panels or wind turbines. In an ideal world, production of this renewable power will be made-to-measure, however the world is not always an ideal place, and wind turbines produce most electricity when there is a lot of wind. In the same way, solar power systems are especially effective when the sun is shining, producing lots of electricity that cannot always be used right away, thus requiring it to be stored. Storage can undertaken with the aid of container-housed battery banks, and it's also possible to store electricity in the form of fuel. In an electrolyzer, water is broken down into its components of hydrogen and oxygen by means of electricity. The hydrogen is stored in special buffer tanks to provide electrical or mechanical energy in a fuel cell or hydrogen combined heat and power plant when it is needed. Also, heat can be extracted if required. A smart cycle – and entirely zero-carbon if the electricity is generated sustainably.
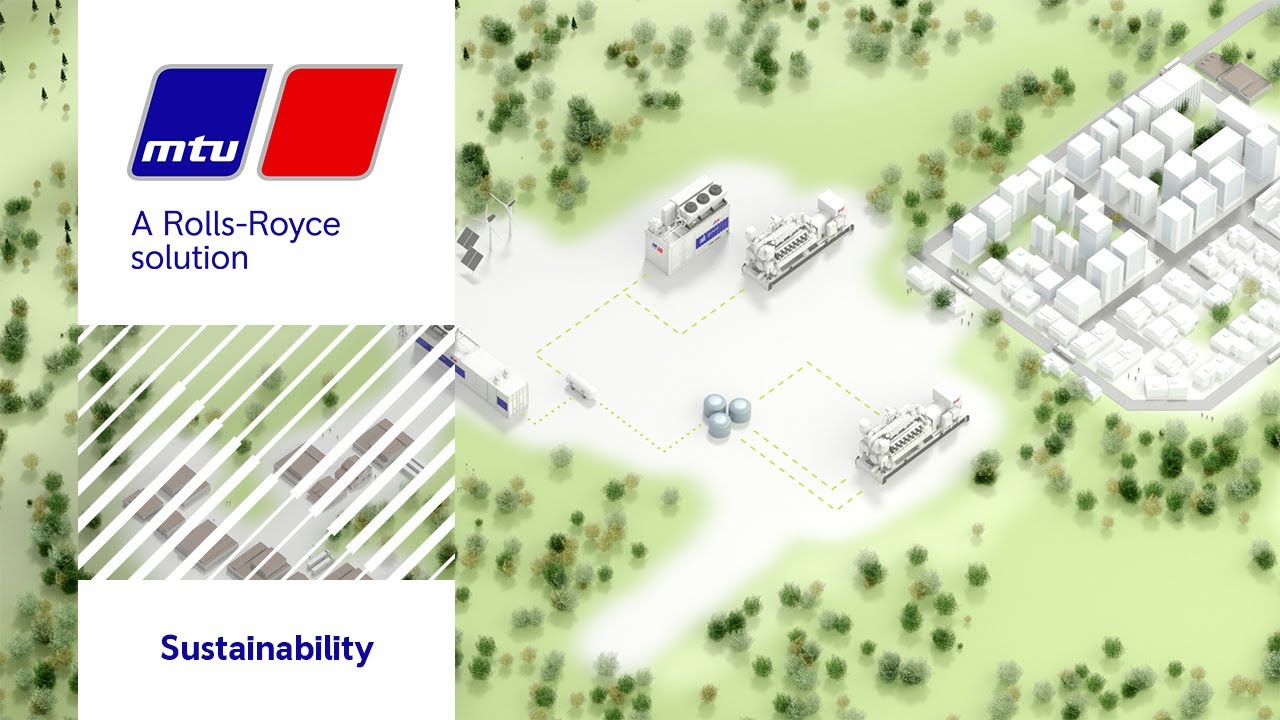
The mtu H2 ecosystem for CO2 neutral fuels
Green hydrogen with mtu electrolysers
High-performance electrolysers are one of the key components of the energy transition. They produce green hydrogen, which can be used as fuel both for fuel cells and combustion engines and further processed into synthetic fuels. mtu electrolysers are equipped with polymer electrolyte membrane (PEM) stacks from Hoeller Electrolyzer. These can produce hydrogen significantly more efficiently and thus at a lower cost than other stacks available on the market.
In the electrolysis of hydrogen, water is placed under DC voltage, producing hydrogen at the negative pole and oxygen at the positive pole. The electrochemical reaction takes place between plate-shaped electrodes separated by membranes. Hundreds of cells, one above the other and pressed together, form a stack, the core of an electrolyzer.
Power-to-X: electricity turned into fuel
The future for internal combustion engines can be found in synthetic fuels produced using what is known as the power-to-X process. Through this process, excess electricity generated from renewable sources such as wind, sun, and water, is converted to hydrogen through electrolysis. This hydrogen can then be used on its own as a fuel, or to produce almost any gaseous or liquid fuel with the addition of CO2 and/or nitrogen from the air. These include substances such as synthetic e-methane, e-methanol, e-ammonia or even e-diesel.
Alternative and sustainable fuels
The future of fuels is diverse. But not every fuel is suitable for every application. Energy density, availability and infrastructure play a major role in selection.
E-Hydrogen
Hydrogen is produced by a process of electrolysis applying electrical current to water. If this electricity comes from renewable sources, the product is referred to as green hydrogen. The fuel contains no carbon, so no CO2 is released during combustion, the only by-product being – once again – water. However, hydrogen has a low energy density – if the necessary tank infrastructure is taken into account. It must be stored under high pressure or in liquid form at very low temperatures (minus 253 degrees Celsius). Because of this, we see its greatest opportunity for use in fuel cells and stationary combustion engines for power generation. As early as 2022, our gas engines will be able to run on 25% hydrogen, and we expect to increase this percentage steadily until we reach 100% hydrogen operation.
E-Methanol
Methanol can be produced carbon-neutrally using the Power-to-X process. Here, hydrogen is synthesized with CO2 in methanol. The energy density of methanol is high in comparison with hydrogen, and since the fuel is liquid at room temperature, it is very convenient to store and transfer. Even existing infrastructure can continue to be used in many cases. Unlike ammonia, methanol is not highly-toxic and is environmentally safe. Methanol can be used not just in diesel and spark-ignition combustion engines, but also in conjunction with zero-emission fuel cells. We're working on both products.
E-Methane
Green hydrogen can be processed further into e-methane by using carbon dioxide from the air or from biogenic sources. This means that existing mtu gas engines can continue to be used carbon-neutrally. For mobile applications, the fuel is usually liquefied into e-LNG (liquid natural gas) to reduce its volume for transportation and storage. Gas engines are used for a variety of applications today and – if the e-methane is produced using renewable electricity – they can run carbon-neutrally as well. However, methane (or e-methane) fuel is not combusted completely in certain load ranges (part-load operation and acceleration phases), allowing some of it to enter the exhaust gas unburned (methane slip). Since methane is harmful to the climate, one challenge in the development of gas engines is to keep methane slip to a minimum.
E-Ammonia
Green ammonia can also be produced carbon-neutrally using the Power-to-X process. Here, green hydrogen is catalytically converted to ammonia using nitrogen in the atmosphere. E-ammonia does not emit CO2 during combustion. The fuel already liquefies at minus 33 degrees and also has a higher energy density than liquid hydrogen. However, the nitrogen-hydrogen compound is highly toxic, and any leaks in coastal waters have the potential to cause severe harm to humans and the environment.
E-Diesel
E-diesel is produced from CO2 and green hydrogen mainly via the Fischer-Tropsch process, which is why this fuel is often referred to as Fischer-Tropsch diesel. Compared to fossil diesel, e-diesel contains hardly any sulfur and no aromatics. It also has a higher ignition propensity due to its chemical structure. As a result, it burns more cleanly and is better suited to storage. The energy density is almost comparable to that of fossil diesel. In the coming year, the engines of our main 2000 and 4000 series will be released for e-diesel and other fuels of the EN 15940 standard. Some engines, for example for power generation or industrial applications, have already been approved.
Significant CO2 savings: second-generation biofuels for mtu engines
Even today, our mtu Series 4000 and Series 1600 engines for power generation are approved to run on synthetic paraffinic diesel fuels in accordance with EN 15940, which includes the biofuel HVO. In 2023, approval for Series 2000 and Series 4000 engines in key applications is expected as well. HVO is one of the first commercially available fuels that can be used to power internal combustion engines in a significantly more climate-friendly way. When the fuel is burned in mtu engines, up to 90% fewer greenhouse gas emissions are produced than when fossil diesel is burned. Also, our combined heat-and-power plants can already run on biogas to generate clean heat and power. By leveraging the emission-reducing possibilities of synthetic fuels in our existing and future engines, we will be well on our way to achieving our 2030 target of reducing total emissions by 35% as compared to 2019.
Related content

Connect with Us